Author Affiliations: Instituto Politecnico Nacional, Centro de Biotecnologia Genomica Nacional, Reynosa, Mexico (R. Palacios-Santana, L. Wei, NA Fernandez-Santos, MA Rodriguez-Perez, JG Estrada-Franco); Texas A&M University, College Station, Texas, USA (NA Fernandez-Santos); Secretaria de Salud de Tamaulipas, Ciudad Victoria, Mexico (S. Uriegas-Camargo); University of Texas Medical Branch, Galveston, TX, USA (NL Mendell, DH Bouyer)
The spotted fever group (SFG) rickettsiae, which are transmitted primarily by tick bite to rodents, dogs, wildlife, and people, comprise a phylogenetically distinct clade of the family Rickettsiacae, encompassing approximately 30 species. SFG rickettsioses are a major cause of febrile illness worldwide. This group includes Rickettsia rickettsii, the bacterium that causes Rocky Mountain spotted fever (RMSF), a major pathogen of public health concern linked to severe hemorrhagic disease in humans. SFG rickettsiae are maintained throughout a tick-vertebrate host cycle; humans are accidental hosts (1). The arthropod-borne typhus (TG) group includes R. typhithe causative agent of murine or endemic typhus, and R.. prowazekiithe pathogen responsible for epidemic typhus (2). R. typhi is supported in enzootic cycles by small mammals, such as rats (Rattus spp.) and opossums (Didelphis spp.), and by ectoparasites such as the rat flea (Xenopsylla cheopis) and the cat flea (Ctenocephalides felis) (3,4). Direct transmission of TG rickettsiae to humans occurs through cross-contamination between mucous or skin abrasions and Rickettsia contained in flea excrement (R. typhi) or human body lice (R. prowazekii) or simply by inhaling contaminated dust (4). However, a sylvatic cycle of R. prowazekii in the United States has been associated with ectoparasites of flying squirrels (Glaucomys volans) (5). The clinical manifestations of TG rickettsioses begin with a sudden onset of fever and other nonspecific symptoms including severe headache, myalgia, arthralgia, nausea and vomiting.
Between 2013 and 2022, a total of 2,232 cases of RMSF were reported in 5 of the 6 contiguous northern Mexican states in the United States (6). Endemicity of TG rickettsiae in southern counties of Texas in the transboundary Rio Grande area of the United States (4) and seropositivity among blood donors in Mexico City (7) and Yucatan (8), Mexico, indicate the need to assess the presence of TG rickettsiae in this area. We report a cross-sectional serological study of SFG and TG rickettsiae conducted in domestic dogs and humans in Reynosa, an urban city in northeastern Mexico. Based on previous research in this city (1), we hypothesized that dogs are sentinels of the infectious disease circulation, as has been reported elsewhere for rickettsiae (9,ten), and are therefore useful in surveillance approaches aimed at monitoring rickettsial disease in humans.
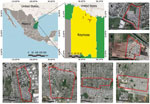
We tested 106 dogs from 71 households in 6 suburban neighborhoods of Reynosa between March 13 and July 4, 2022 (Figure). We collected individual signed informed consents from dog owners and recorded data on the sampled dogs, including their vaccination history, sex, age and breed. We administered the rabies vaccine to unvaccinated dogs. We recruited all household members (n = 36) with dogs seropositive for SFG or TG, 16 of whom provided blood samples for further analysis. We collected and processed blood samples (≈3–5 mL, dog or human) according to previously reported methods (1). We centrifuged the blood at 3000× g for 10 min and serum samples stored at -80°C until further processing. We tested serum samples for IgG against R. amblyommatis, R.parkeri, R.rickettsiiAnd R. typhi by indirect immunofluorescence test as previously described (11). We recorded serum samples with reciprocal iIgG titers ≥64 or greater as positive titer and determined the endpoint of these samples.
Of the dogs sampled, 5 (4.71%, 95% CI 0.68% – 8.75%) were positive for GFS. Rickettsia IgG and 4 (3.77%; 95% CI 0.15% – 7.4%) were TG positive Rickettsia IgG antibody (Annex Table 1). To further test whether HIV-positive dogs were sentinels of rickettsia exposure, we collected blood samples from dog owners and their families and asked them about recent tick bites and clinical history ( previous 3 months) associated with signs of rickettsial disease (Annex Table 2) (12). Testing revealed that 5 (32.3%; 95% CI: 1.84% to 53.9%) of 16 serum samples obtained from owners of households with positive dogs were positive for GFS. Rickettsia IgG under identical experimental conditions (Annex Table 1). Tamaulipas Health Services is conducting a follow-up survey for medical evaluation and treatment of SFG HIV-positive individuals.
RMSF was described in Mexico in 1943. We report the discovery of anti-SFG antibodies Rickettsia in Reynosa, Mexico in 2022. Serological evidence of exposure of a domestic dog to R. typhi in Reynosa warrants further investigation, especially given the endemicity of murine typhus in contiguous Hidalgo County, Texas, USA, which involved more than 1,000 human cases reported between 2008 and 2019. Our results validate the theory that dogs can be sentinels of human diseases and demonstrate their aid in inferring the temporal and spatial dynamics of certain diseases in regions associated with their vectors (12).
The metropolis of Reynosa is an industrial center and an area of cross-border migration of reference between different demographic groups comprising the Reynosa-Texas-United States region, known as the Rio Grande Valley. It is a ≈110 km strip of land with ≈3 million permanent residents, with a large floating population fluctuating between several hundred and several thousand. The entire cross-border region between Mexico and the United States extends over 3,141 km, and more than 286 million crossings are documented each year (13).
Historical outbreaks of RMSF have been documented along the Mexico-US border region, with mortality rates of 30–80% (12). In 2019, an RMSF seroprevalence of 6% (95% CI 4.68%-7.46%; 69/1,136 cases) in dogs was reported in 14 Mexican border towns in the states of Baja California, Sonora and Coahuila (14), which is consistent with the seroprevalence of our study (4.71%; 95% CI 0.68% – 8.75%). Human cases of murine typhus have been reported primarily in southeastern Mexico, but rarely in northeastern Mexico. However, endemicity in the Rio Grande Valley has been widely reported on the US side of the border (4). In 2022, 23.9% (95% CI 16.9%-31.0%; 34/142 samples) of human serum samples were positive for R. typhi IgG in Yucatan, and 15% of them were positive for R.rickettsii (15). Similarly, the first screening of HIV-positive Rickettsia IgG in dogs has identified putative human cases, indicating that this surveillance strategy is effective (15).
Most dogs roam freely in relatively squalid conditions in our study areas. In this context, the evidence of TG Rickettsia antibodies in dogs did not match the same results in their owners. On the contrary, owners of SFG seronegative dogs suggest the possibility that the owners acquired the infection away from their residence and their pets. Although the antibody titers of 3 of 5 SFG-reactive human samples suggest R.parkeri as a presumptive agent, without paired serum samples, other SFG rickettsiae cannot be ruled out. Due to the cross-reactivity of rickettsioses observed by immunofluorescence assay, further studies using cross-uptake techniques to clarify the causative etiologic agent(s) are warranted. This pattern of cross-border transmission appears to be repeated along Mexican cities abutting the U.S. border, where the brown dog tick (Rhipicephalus sanguineus) infestations are known to be endemic.
Our results suggest that the epidemiological fabric of the region is strongly affected by the high incidence of SFG and TG rickettsioses. We hypothesize that improving the urban environment, using a One Health approach, together with integrated vector control management of ectoparasites (e.g. tick collars for dogs, trapping of ticks, miticide spraying) would be of utmost importance in reducing the spread of rickettsial diseases in the regions. as our study area. Evidence of rickettsia TG exposure in domestic dogs in this study highlights the need for further surveillance to determine vector phenology and transmission cycle in the region. In conclusion, surveillance and control of SFG and TG rickettsia, through standard and novel approaches, is urgently needed in areas along the northeastern border between Mexico and the United States.
Mr. Palacios-Santana is a graduate student at Centro Biotecnologia Genomica-IPN and works on vector-borne diseases, with an emphasis on Rocky Mountain spotted fever.